I was talking to a group of my junior registrars the other day about cardiac physiology, and I was struck by how much trouble they seemed to have relating electrolytes to the QRS complex. We sat down and nutted it all out which left everyone happy. The key to understanding the effects of electrolytes is to understand the cardiac action potential, and how it relates to the QRST complex seen on an ECG. Below is a brief summary of our conversation.
Think for a moment about the basic physiology of the cardiac action potential.
I want you to forget for a moment about all those messy classifications of phase 0 through to phase IV, and don’t for a moment let the Nernst Equation enter your mind.
Just remember that the charge of a cellular membrane depends on 4 ions:
1) Sodium (Na+)
2) Calcium (Ca++)
3) Potassium (K+)
4) Chloride (Cl–)
Chloride doesn’t play much of a role in the cardiac potential so you can forget about it. (“Thank goodness!” I hear you sigh). Remember that Sodium and Calcium predominantly exist in the extracellular space, so when the membrane of a cell becomes permeable to their passage they rush into the cell and give it a large +ve electrical charge. Potassium has the opposite effect.
Got that straight in your head? Good.
The Action Potential in Action
Phase 0
Normally, cardiac myocytes sit with a resting membrane potential of somewhere between -85mV and -95mV.
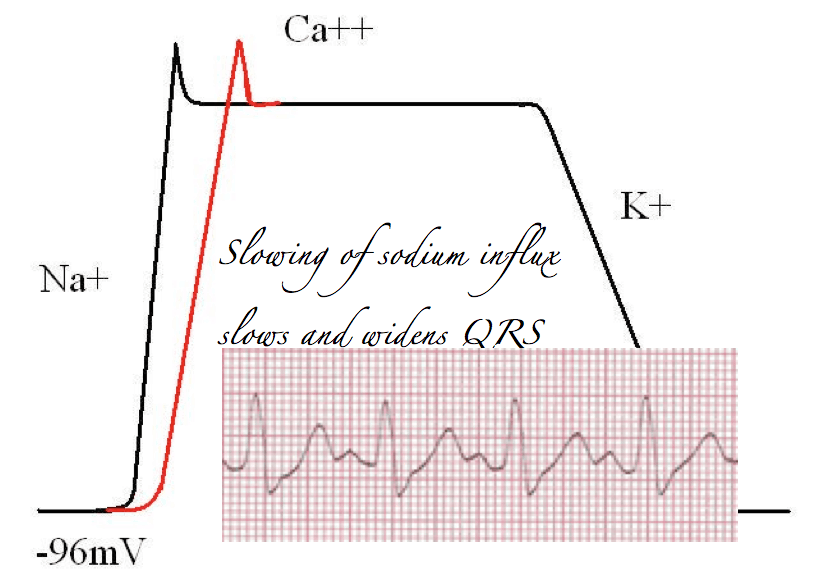
When the cells become excited sodium channels open. There is a massive influx of sodium (remembering it’s a cation) into the cells and a resulting large positive shift in the resting membrane potential.
This creates a large positive electrical vector. Such a shift shows up as a large deflection on any device designed to measure electrical current, such as an ampere-meter or an ECG recorder.
Phase 2.
I’ve skipped “phase 1” of the action potential, because it’s got a lot of technical details that aren’t really relevant to what we’re talking about.
Once the sodium channels have finished working their magic, calcium channels take up the cudgel and open up. This means that the large positive charge of the cardiac membrane is essentially maintained at level. This manifests as a flat line, or plateau phase. Because there is no major change in the charge of the membrane, there is nothing for our device to measure, and this phase will manifest as a straight line.
Phase 3.
Eventually, calcium influx slows, and potassium channels open, repolarising the heart. This lowers the membrane potential back to its resting levels, and manifests as a large electrical change that can be measured on a current recorder.
The ECG is the sum of millions of myocytes.
Once you’ve got your head around that concept, understanding the ECG in relation to the action potential is relatively easy.
When the cardiac myocytes contract, millions together at once, there is a large change in the electrical field of the heart. Of course, they don’t all contract completely simultaneously – it normally takes them about 0.12 of a second…
In other words, the QRS complex is the product of sodium channel opening in cardiac myoctyes.
If you’re still following, you’ll realise  that after the sodium channels have finished their business and the calcium channels open, this results in a long period of non-measurable electrical stability, straight after the QRS complex.
that after the sodium channels have finished their business and the calcium channels open, this results in a long period of non-measurable electrical stability, straight after the QRS complex.
That’s right, I’m talking about the ST segment. It’s the “calcium” bit of cardiac conduction.
The final part should be obvious. As we’ve already described, the influx of K+ when the right channels open causes another large electrical spike, manifesting on the ECG as a T wave.
This is why medical students are often told to remember that the T wave is “full of K+“.
So, what does this have to do with electrolytes?
One of the hardest things to do is to remember what the effects of different electrolyte imbalances are on the ECG. But, if you remember the basic physiology of electrolytes, it’s dead simple.
Block sodium channels, and the rate of rise of sodium influx is slowed. This means muscle contractions will take longer and prolong the QRS. Thus, particularly from a toxicology perspective, prolongation of the QRS, should alert the astute reader of an ECG to the presence of sodium channel blockade.
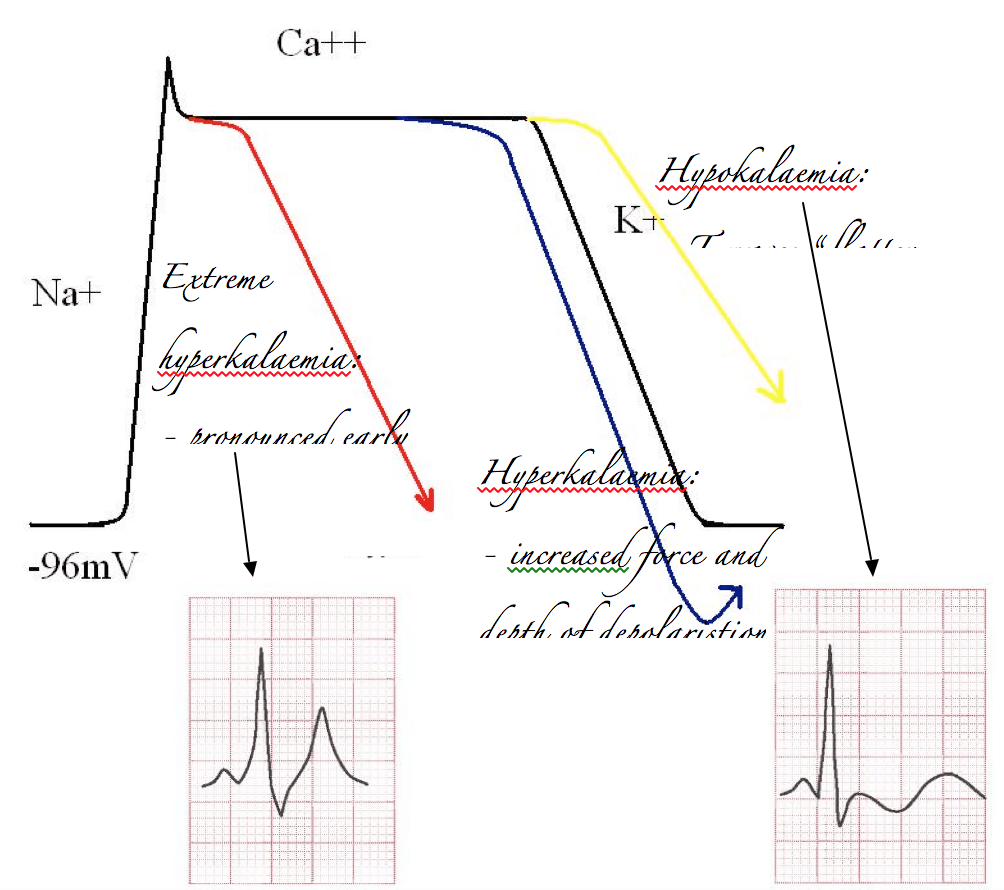
Alternatively, if you’ve got too much potassium on board it’s easy to predict that the influx of potassium will be more impressive than it otherwise would. Therefore the repolarisation curve will be steeper than normal, and overshoot. This will increase the size and volume of the electrical deflection measured on the ECG, which is of course the T wave. In extreme cases this will cause very early repolarisation, and at the same time an increase in basal K+ exchange across the membrane will slow the rate of sodium induced depolarisation. A sinusoidal waveform develops.
The opposite effect occurs with hypokalaemia. Depolarisation is less impressive, and T waves “flatten out”.
What about calcium?
Calcium is a bit trickier. Classically hypercalcaemia is taught to shorten the QT interval. This is not quite what you’d expect from more calcium. You’d be forgiven for thinking that more calcium would prolong the plateau phase and increase the QT interval. (I should point out in the real world that the association between calcium and QT shortening is dubious at best.) But, you need to remember a couple of things:
– Calcium is a positive inotrope, and increased contractility shortens the QT
– Calcium transport affects potassium transport. More calcium causes bigger shifts in potassium, and may cause earlier depolarisation.
That all seems a bit long winded!
And it is. If you’ve read this far, congratulations! Remember:
Sodium is the primary ion affecting the QRS complex.
Potassium is the primary ion affecting T waves
If there is a QT abnormality, you should check a serum calcium!
Luke Lawton









Thankful nursing student here! Please follow up with an explanation on calcium 🙂
God bless you .
Coming soon.
If you want a detailed discussion and are a Cardiac Bootcamp Course Member, just go to the electrolytes and the Heart section.
https://www.emcore.com.au/self-study-course.html
Electrolytes and the Heart – Resus
[url=http://www.gg082nmw5732jm4p0fe84654c6kwup8ss.org/]udzrwlowfyp[/url]
adzrwlowfyp
dzrwlowfyp http://www.gg082nmw5732jm4p0fe84654c6kwup8ss.org/
What a material of un-ambiguity and preserveness of precious knowledge regarding unexpected emotions.
I appreciate, result in I found exactly what I was taking a look for.
You have ended my 4 day long hunt! God Bless you man. Have a nice day.
Bye
My homepage; Max Melt Keto Ingredients
I’m gone to convey my little brother, that he should also pay a quick visit this blog on regular basis to
get updated from most recent reports.
Also visit my web page; Extreme Keto EFX Review
Wow, amazing blog layout! How long have you been blogging
for? you make blogging look easy. The overall look of your website
is fantastic, as well as the content!
my blog :: Max Melt Keto Ingredients
Heya i’m for the first time here. I came across this board and I find It truly useful & it helped me out much.
I am hoping to present one thing back and help others like you helped me.
Here is my blog: Extreme Keto EFX Pills (http://www.mhes.tyc.edu.tw)
Do you have a spam issue on this website; I also am a blogger, and I was wanting to know your situation; many of us have created some nice practices and we are
looking to swap strategies with other folks, be sure to shoot me
an email if interested.
Feel free to visit my blog … GrownMD CBD Review
Hi every one, here every person is sharing these know-how,
thus it’s good to read this website, and I used to visit this
weblog daily.
Also visit my page :: Pure CBD Softgels Review (http://www.hltkd.tw/hl4/userinfo.php?uid=191213)
Hi, I do believe this is an excellent blog. I stumbledupon it 😉 I will return once again since I bookmarked it.
Money and freedom is the best way to change,
may you be rich and continue to guide others.
my web blog; TestoXmen Review (http://www.mhes.tyc.edu.tw)
Real excellent info can be found on web blog.
Feel free to surf to my site: Bioneo Farms Pure CBD (http://www.aniene.net)
Utterly indited content material, Really enjoyed reading through.
my web blog; Tundra Breeze Portable Air Conditioner
you are actually a good webmaster. The site loading
pace is incredible. It kind of feels that you are doing any unique trick.
Also, The contents are masterpiece. you have done a fantastic task on this subject!
Also visit my web page UltraXTend (Mazie)
I’ve been surfing online more than 4 hours today, yet I never found any interesting article like yours.
It’s pretty worth enough for me. In my view, if
all web owners and bloggers made good content as you did, the internet will be much
more useful than ever before.
Feel free to surf to my homepage – Terra Xtract CBD
Thanks very interesting blog!
Here is my page :: Ultra Quick Keto Burn (http://www.hltkd.tw/hl4/userinfo.php?uid=190193)
Very good website you have here but I was wondering if you knew of any user
discussion forums that cover the same topics discussed here?
I’d really like to be a part of group where I
can get advice from other knowledgeable people that share the
same interest. If you have any suggestions, please let
me know. Bless you!
My web-site: Erectesto XL (http://www.comptine.biz/modules.php?name=Your_Account&op=userinfo&username=SiobhanAzm)
I was curious if you ever considered changing the structure of your site?
Its very well written; I love what youve got to say. But maybe you could a
little more in the way of content so people could connect
with it better. Youve got an awful lot of text for only having one or
two images. Maybe you could space it out better?
My web blog: Bioneo Farms CBD Reviews (http://www.chubbychannel.com/)
I just could not go away your site before suggesting that
I actually loved the usual information a person supply
for your guests? Is gonna be again incessantly in order to check
out new posts
Today, while I wass at work, my cousin stol myy iphone and tested to
see if it can survive a 25 foot drop, just so she can be
a youtube sensation. My apple ipad is now broken and shee has 83 views.
I know this iis completely off topic but I had to share
it with someone!
My website; cần mua máy game bắn cá
What’s up, its nice post concerning media print, we all be aware
of media is a wonderful source of facts.
Alibaba Website Lock Pin Latch
Gucciグッチイヤリングコピー
portable public toilet
Tiffanyティファニーイヤリング販売店
Attic Exhaust Fan Replacement
スーパーコピーバッグ
Chanelシャネル靴販売店
Wood Dryer
gotta go porta potty
Hermesエルメスブレスレットコピー
1 Function Hospital Bed
ブランドコピー専門店
Live Catch Cage Traps
スーパーコピーブランド
15hp Gasoline Engine
ブランド帽子コピー代引き
スーパーコピーブランド
Soundproofing Blanket
Organic Synthetic Extractants And Cleaning Agents
ブランド時計コピー
ブランド時計コピー
Iron On Transfer Paper Laser
Fast Charging Car Station
ブランド時計コピー
12mm Film Faced Plywood
ブランドコピー専門店
ブランドコピー専門店
Airtight Ziplock
Raw Paper Slitter for Straw