An 84 year old male has been brought into the emergency department. He has been found at home, confused, by a visiting neighbour. On arrival to the emergency department, the patient’s observations are: GCS 14/15, T 38.6 C, Pulse 143bpm and saturations of 94%. His clinical examination is normal apart from right basal crepitations. His initial BSL on fingerpick comes back at ‘critical high’.
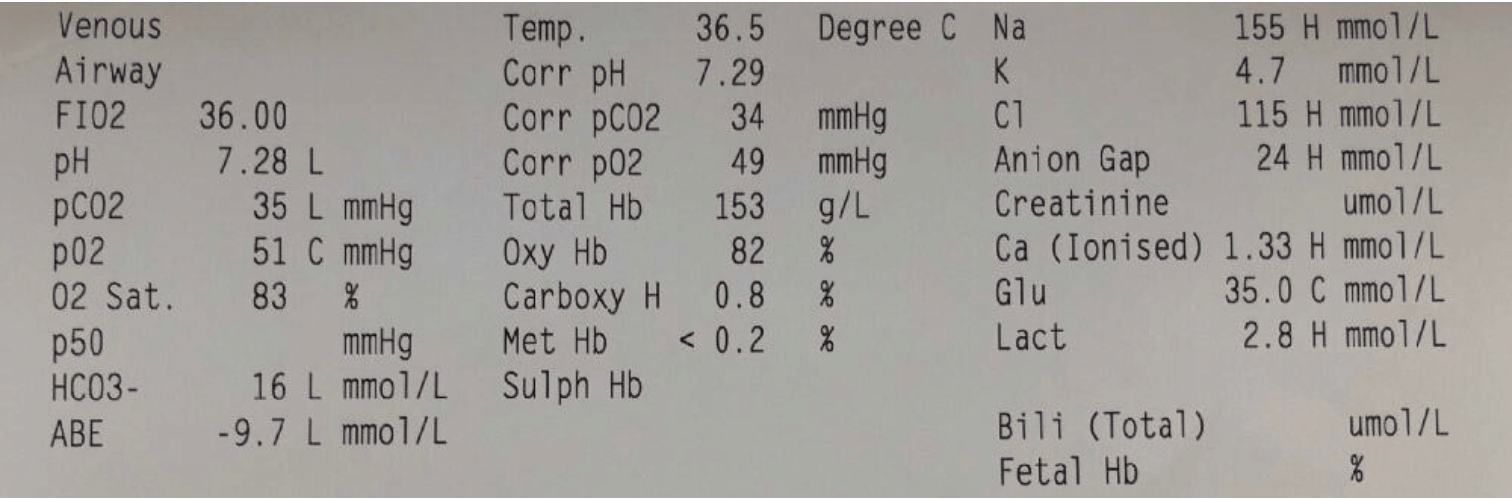
A venous gas shows the following:

The patients Ketones were 6.6 and Osmolality was 350
You working diagnosis is probable pneumonia causing hyperglycaemia and ketosis.
If you are a registrar and even if you are not have ago at the calculations:
Describe and Interpret The Venous Gas
Here is my simple approach, the same one we teach in the Fellowship Course:
Primary Disorder is Metabolic Acidosis
Co2 Compensation: Expected is (1.5 x HCO3) + 8 +/- 2 = 32 +/- 2 ie 30-34 = Compensated
Anion Gap= 155-(115+16) = 24= High Anion Gap Metabolic Acidosis(HAGMA)
Delta Ratio: change in AG/change in HCO3 = 24-12/24-16=1.5 = pure HAGMA
Glucose is high at 35
Lactate is high probably second to hypo perfusion.
Corrected K is 5.5 mmol/L
Corrected Na = Na +(Glc-5)/3 = 165
Outline your approach to fluid resuscitation in this patient
This is a severely hypernatraemia patient that also has a high osmolality. We have no history on the patient in terms of whether he has chronic hypernatraemia, but have to assume that this is an acute event. When managing all sodium abnormalities, high or low remember to GO SLOW! The rapid reduction in tonicity can cause significant problems. A recent blog on Hypernatraemia went through all the calculations required.
Assuming a weight of 70kg.
Total body water(TBW) = 0.5 x 70 = 35
Total Water deficit = TBW x 1 – (Na desired/Na actual) = 35 x (1-(140/165)) = 5.3 Litres of fluid
What fluid and how?
In most patients we must not lower the Na by more than 1-2mmol/L / hour
In this case we would not use 5% Dextrose, given the glucose is already high. We could go directly to 4.5% NSaline and give 500mL. Is that all we can give?
What would happen if we gave 1L of Normal Saline?
Change in Na with 1 L Saline = (Na in fluid-serum Na)/TBW+1 = 154-165/35+1 = -0.3mmol/L
ie the Na would be lowered by 0.3mmol/L given that the fluid infused and the serum levels are both high. There would be an effect on the osmolality however given the small drop in sodium, this would not be expected to be high.
So in a situation like this, NSaline can be used and then switched over to 4.5%NSaline. The key is to remeasure the Na regularly as patients vary in their responses to these fluids.
Go to the blog for a full review of Hypernatraemia and Hyponatraemia.